
"Unlock your desired look with personalized cosmetic surgery or medical spa services. Explore our range of procedures tailored to your unique needs and goals."
Dr. Wang

Reveal timeless radiance through expert Face Sculpting procedures, tailored to enhance your unique beauty

Achieve natural, balanced results with expert care and personalized breast enhancement procedures
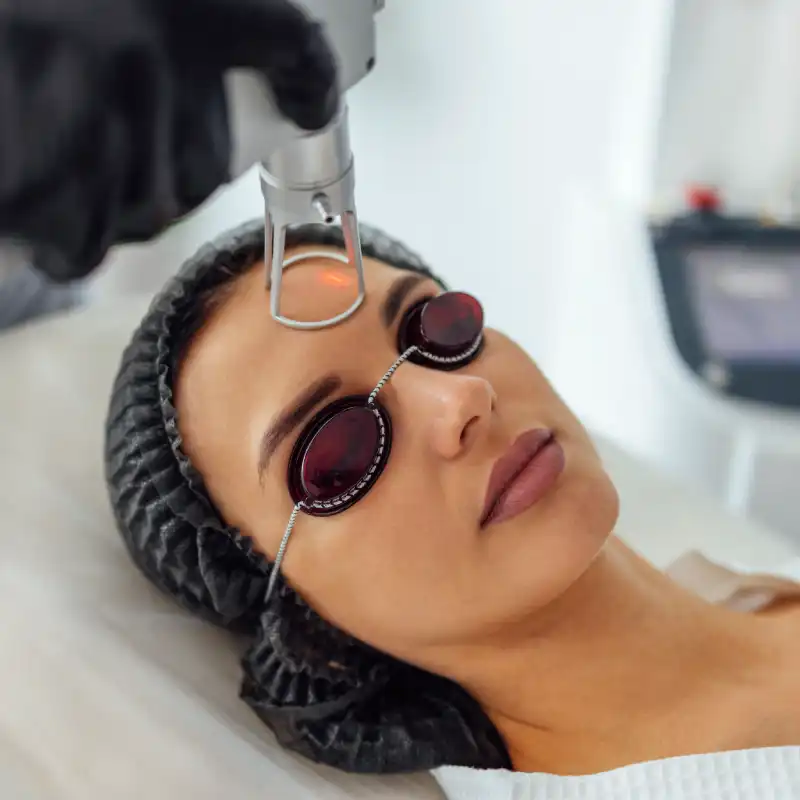
Transform your skin with rejuvenating treatments, unlocking a luminous complexion and renewed sense of vitality

Sculpt your ideal silhouette with precision Body Contouring, addressing stubborn areas to achieve harmonious proportions
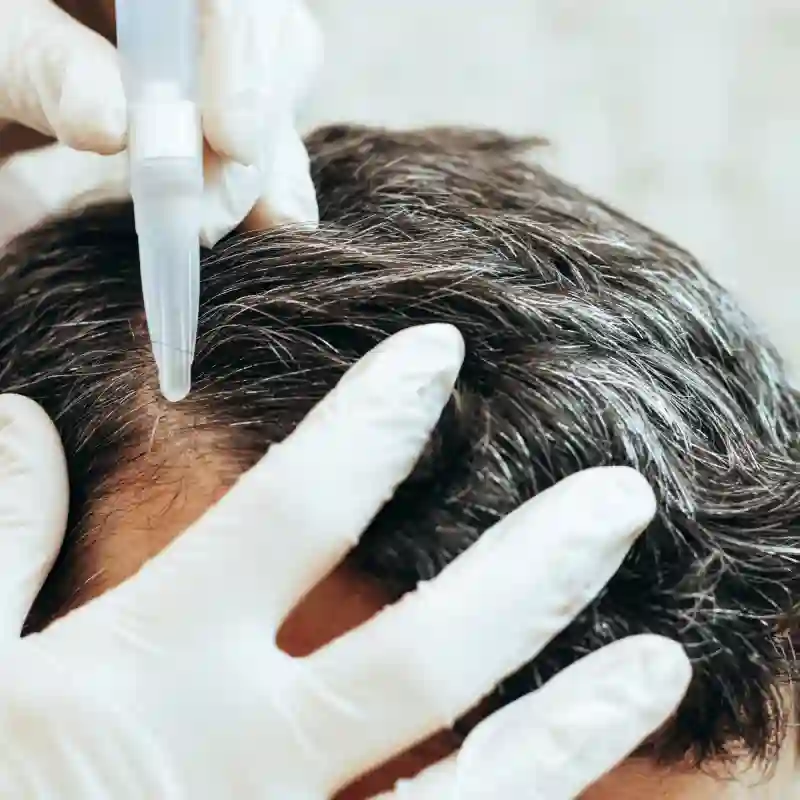
Rediscover confidence with Hair Restoration, where art meets science to restore and enhance your natural hairline

BREAST LIFT | BREAST AUGMENTATION | TUMMY TUCK
“I was able to see that my stomach was flat. When he took off the bra top, I could see how full my breasts were. The scar is so minimal and thin, you can barely see it..”

ACCUSCULPT™ | PASADENA
"I was so disappointed from my first liposuction that I didn't think it was possible to have it look this good. I wish I had found Dr. Wang from the start."

UPPER & LOWER EYELID SURGERY | BOTOX
"I have total confidence in Dr. Wang. Now I look like guys my age should. I feel better when I see myself in the mirror, and my wife says I look at least 5 years younger. If I was going to get anything else done, I wouldn't shop around-I'd go straight to Dr. Wang."

BREAST LIFT | BREAST AUGMENTATION | TUMMY TUCK
“I was able to see that my stomach was flat. When he took off the bra top, I could see how full my breasts were. The scar is so minimal and thin, you can barely see it..”

ACCUSCULPT™ | PASADENA
"I was so disappointed from my first liposuction that I didn't think it was possible to have it look this good. I wish I had found Dr. Wang from the start."

"Unlock your desired look with personalized cosmetic surgery or medical spa services. Explore our range of procedures tailored to your unique needs and goals."
Dr. Wang
Research your surgeon, understand the risks, have realistic expectations, and ensure proper post-operative care for optimal results

A great plastic surgeon meets the highest professional and personal standards with impeccable credentials.

Learn about nature’s healing curve, the emotional roller coaster and advice from Dr. Wang.

After your consultation, our patient consultants will present a quote for the cost of the treatment option you choose.

Some of the most common questions Dr. Wang gets from his plastic surgery patients.
Take the first step towards a more confident you. Reach out to Wang Plastic Surgery & Med Spa now, and let our experts guide you on your beauty-enhancing path.
Contact Wang Plastic Surgery & Med Spa, and let our skilled team guide you toward achieving your beauty goals
















Board certified plastic surgeon Dr. Stewart Wang specializes in Pasadena breast augmentation as well as breast enhancement, hair transplant, face lift, laser hair removal, eyelid surgery (blepharoplasty), nose surgery (rhinoplasty), abdominoplasty (tummy tuck) and breast lift. He has cosmetic surgery offices and med spas in Upland, Pasadena, Glendora and Santa Ana and serves Southern California including La Verne, San Dimas, Chino Hills, Diamond Bar, Yorba Linda, Riverside, Rancho Cucamonga, Brea, Fullerton, Orange, Tustin, and Irvine.